How to Know if a Molecule Has Resonance
If a molecule is unstable in some way it will want to react. Since the molecular formula is O 3 we know there are 18 valence electrons oxygen has six valence.

Resonance Structures Stability Based On The Octet And Formal Charge Chemistry Octet Rule Inductive Effect
4 When two or more structures can be drawn the one with the fewest total charges is the most stable.

. If the steric number is 4 it is sp3. Resonance structures must all have the correct. 1 The same molecular formulas.
3 The same atoms connected together. Whenever you can draw two or more. Stability and reactivity generally have an inverse relationship.
I hope someone can help me out. There are a few rules that must be followed when determining resonance of compound. A molecule can have resonance structures when it has a lone pair or a double bond on the atom next to a double bond.
The two oxygens are both partially negative this is what the resonance structures tell you. To find the resonance structure of ozone we will draw the lewis structure of ozone. The overall electronic structure of the molecule or ion is given by the weighted average of.
Answer 1 of 4. 1 A lone electron pair containing polyvalent atom is present in the. How do we know when to draw a resonance structure.
As you can see the carbon with two hydrogens has five bonds 10. Resonance only occurs when a molecule has at least one double bond. A molecule shows resonance if the following conditions are fulfilled.
In these pi orbitals the electrons would be scattered over. Here are the 4 signs we can lo. To know when we.
This movement of the electrons is called delocalization. Resonance arises when more than one valid Lewis structure can be drawn for a molecule or ion. Resonance occurs in species for which there are two or more valid Lewis.
Resonance refers to the existence of numerous forms of a compound and is a component of valence bond theory. Of conjucations hence 5. Molecules with only single bonds never show resonance.
If the steric number is 2 sp. A molecule can have resonance structures when it has a lone pair or a double bond on the atom next to a double bond. If you cannot spot the carbons with exceeding octet immediately add the invisible hydrogens on the bond-line structure.
Owing to the overlap of orbitals resonance exists. In resonance structures the electrons are able to move to help stabilize the molecule. Shown below are the two resonance structures of.
In chemistry terms resonance describes the fact that electrons are delocalized or flow freely through the molecule which allows multiple structures to be possible for a given molecule. If the steric number is 3 sp2. Now you have no.
Resonance structures should have the. So now lets go back to our molecule and determine the hybridization states for all the atoms. For example if the starting molecule has a 1 charge the resonance structure must also have an overall charge of 1.
Double bonds formed from the overlap of 2p orbitals are composed of pi bonds. 2 The same total number of electrons same overall charge. How do you tell if a molecule shows resonance.
Of RS Just observe and calculate no. Multiple charges on atoms CAN exist. How can I determine if a molecule has resonance without having to draw anything if thats even possible.
Whenever you can draw two or more Lewis structures for a. Combine each atom with a single bond to the central atom by contributing one electron from each atom for the bond. Count the electrons in the valence shell to.
While both resonance structures are chemically identical the negative charge is on a different. Yes there is an super easy way to find no. In the example below A is more stable than B.
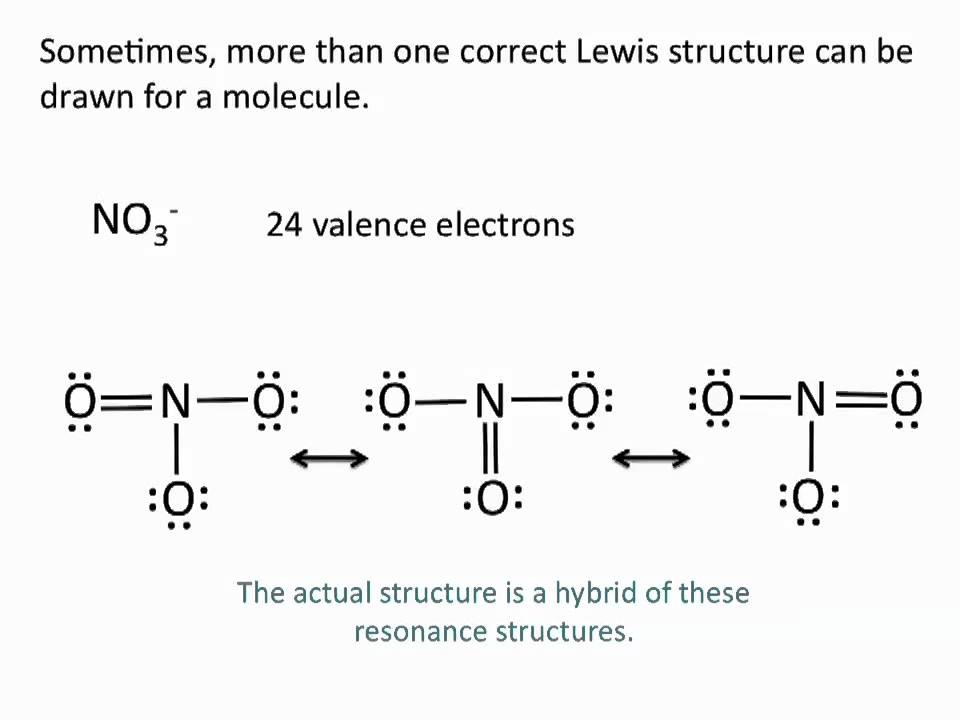
Drawing Lewis Structures Resonance Structures Chemistry Tutorial Youtube Chemistry Science Education Organic Chemistry

Resonance Structures Mcat Electrons Chart

Scn Lewis Structure Thiocyanate Ion Molecules Lewis Math

Curved Arrows In Resonance Structures And Chemical Reactions Chemistry Chemical Reactions Curved Arrow
No comments for "How to Know if a Molecule Has Resonance"
Post a Comment